Empfohlen
We analysed feedback from a series of peer-led lectures for large groups of 100-150 students to determine if students are able to provide high-quality interactive case-based
lectures comparable to standard medical curricula
This was presented as a short communication at Association for Medical Education in
Europe (AMEE) Conference, August 2013 (3M/1) and FRAMPEIK Student Research Conference, October 2013Peer lectures in the medical undergraduate curriculum: a valid tool for lear...

Peer lectures in the medical undergraduate curriculum: a valid tool for lear...UCL Medical Society (Education) 2012-2013
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Andere mochten auch
We analysed feedback from a series of peer-led lectures for large groups of 100-150 students to determine if students are able to provide high-quality interactive case-based
lectures comparable to standard medical curricula
This was presented as a short communication at Association for Medical Education in
Europe (AMEE) Conference, August 2013 (3M/1) and FRAMPEIK Student Research Conference, October 2013Peer lectures in the medical undergraduate curriculum: a valid tool for lear...

Peer lectures in the medical undergraduate curriculum: a valid tool for lear...UCL Medical Society (Education) 2012-2013
Andere mochten auch (20)
Peer lectures in the medical undergraduate curriculum: a valid tool for lear...

Peer lectures in the medical undergraduate curriculum: a valid tool for lear...
Lesson plan problem related to cylinder, cone, and sphere

Lesson plan problem related to cylinder, cone, and sphere
Philosophical Foundation of curriculum (Edam 514 curriculum development)

Philosophical Foundation of curriculum (Edam 514 curriculum development)
Basic curriculum devlopment concept and curriculum devlopment models.

Basic curriculum devlopment concept and curriculum devlopment models.
Ähnlich wie 4.2
Ähnlich wie 4.2 (20)
Periodic table of elements by Muhammad Fahad Ansari 12IEEM14

Periodic table of elements by Muhammad Fahad Ansari 12IEEM14
Mehr von ZB Chemistry
Mehr von ZB Chemistry (20)
Kürzlich hochgeladen
Making communications land - Are they received and understood as intended? webinar
Thursday 2 May 2024
A joint webinar created by the APM Enabling Change and APM People Interest Networks, this is the third of our three part series on Making Communications Land.
presented by
Ian Cribbes, Director, IMC&T Ltd
@cribbesheet
The link to the write up page and resources of this webinar:
https://www.apm.org.uk/news/making-communications-land-are-they-received-and-understood-as-intended-webinar/
Content description:
How do we ensure that what we have communicated was received and understood as we intended and how do we course correct if it has not.Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...Association for Project Management
Mehran University Newsletter is a Quarterly Publication from Public Relations OfficeMehran University Newsletter Vol-X, Issue-I, 2024

Mehran University Newsletter Vol-X, Issue-I, 2024Mehran University of Engineering & Technology, Jamshoro
Kürzlich hochgeladen (20)
Fostering Friendships - Enhancing Social Bonds in the Classroom

Fostering Friendships - Enhancing Social Bonds in the Classroom
Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...
Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf

Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf
Food safety_Challenges food safety laboratories_.pdf

Food safety_Challenges food safety laboratories_.pdf
Basic Civil Engineering first year Notes- Chapter 4 Building.pptx

Basic Civil Engineering first year Notes- Chapter 4 Building.pptx
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf

UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf
Unit-V; Pricing (Pharma Marketing Management).pptx

Unit-V; Pricing (Pharma Marketing Management).pptx
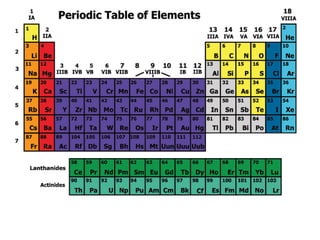
4.2
- 2. Elements are arranged: Vertically into Groups Horizontally Into Periods
- 3. If you looked at one atom of every element in a group you would see…
- 5. The group 2 atoms all have 2 electrons in their outer shells Be (Beryllium) Atom Mg (Magnesium) Atom
- 7. If you looked at an atom from each element in a period you would see…
- 8. Each atom has the same number of electron holding shells . An example…
- 9. The period 4 atoms each have 4 electron containing shells K (Potassium) Atom Fe (Iron) Atom Kr (Krypton) Atom 4 th Shell
- 12. Alkaline Earth Metals Silvery-White Metals Fairly reactive Many are found in rocks in the earth’s crust
- 13. Transition Metals Malleable (easily bent/hammered into wires or sheets) Most are good Conductors of electricity
- 15. Metalloids lie on either side of these “stairsteps” They share properties with both metals and non-metals Si (Silicon) and Ge (Germanium) are very important “semi-conductors”
- 16. What are semiconductors used in?
- 17. Nonmetals Brittle Do not conduct electricity
- 18. Most are Poisonous Fairly reactive Halogens
- 19. Chlorine Gas was used as a chemical weapon during World War I. It was used by the Nazis in World War II.
- 20. Unreactive Gases at room temperature Noble Gases
- 23. Lanthanide Series Actinide Series