What Do Rheumatologists Think?
•
0 gefällt mir•378 views
A recent nationwide survey of rheumatologists has revealed their concerns over the biosimilars approval process and their support of clear regulatory standards for these drugs.
Melden
Teilen
Melden
Teilen
Downloaden Sie, um offline zu lesen
Empfohlen
14. Dr. Thomas Kirchlechner - Sandoz Biopharmaceuticals Development

14. Dr. Thomas Kirchlechner - Sandoz Biopharmaceuticals DevelopmentInternational Federation of Pharmaceutical Manufacturers & Associations (IFPMA)
Weitere ähnliche Inhalte
Andere mochten auch
14. Dr. Thomas Kirchlechner - Sandoz Biopharmaceuticals Development

14. Dr. Thomas Kirchlechner - Sandoz Biopharmaceuticals DevelopmentInternational Federation of Pharmaceutical Manufacturers & Associations (IFPMA)
Andere mochten auch (14)
14. Dr. Thomas Kirchlechner - Sandoz Biopharmaceuticals Development

14. Dr. Thomas Kirchlechner - Sandoz Biopharmaceuticals Development
Biopharmaceuticals (Transforming proteins and genes into drugs)

Biopharmaceuticals (Transforming proteins and genes into drugs)
Pioneering novel approaches to increase access for patients worldwide

Pioneering novel approaches to increase access for patients worldwide
Bio similar- An opportunities or challenge for Indian Company 

Bio similar- An opportunities or challenge for Indian Company
Mehr von Small World Social
Mehr von Small World Social (10)
5 Upcoming Biosimilar Events You Don't Want to Miss

5 Upcoming Biosimilar Events You Don't Want to Miss
The Complexities of Social Media, kids and the Classroom

The Complexities of Social Media, kids and the Classroom
Overcoming the camel's hump with digital literacy: how Web 2.0 is changing ed...

Overcoming the camel's hump with digital literacy: how Web 2.0 is changing ed...
Kürzlich hochgeladen
❤️Call girls in Jalandhar ☎️9876848877☎️ Call Girl service in Jalandhar☎️ Jal...

❤️Call girls in Jalandhar ☎️9876848877☎️ Call Girl service in Jalandhar☎️ Jal...chandigarhentertainm
Kürzlich hochgeladen (20)
Enjoyment ★ 8854095900 Indian Call Girls In Dehradun 🍆🍌 By Dehradun Call Girl ★

Enjoyment ★ 8854095900 Indian Call Girls In Dehradun 🍆🍌 By Dehradun Call Girl ★
VIP Call Girl DLF Phase 2 Gurgaon (Noida) Just Meet Me@ 9711199012

VIP Call Girl DLF Phase 2 Gurgaon (Noida) Just Meet Me@ 9711199012
Tirupati Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

Tirupati Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
Bareilly Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

Bareilly Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
(Sonam Bajaj) Call Girl in Jaipur- 09257276172 Escorts Service 50% Off with C...

(Sonam Bajaj) Call Girl in Jaipur- 09257276172 Escorts Service 50% Off with C...
Call Girls Chandigarh 👙 7001035870 👙 Genuine WhatsApp Number for Real Meet

Call Girls Chandigarh 👙 7001035870 👙 Genuine WhatsApp Number for Real Meet
Sambalpur Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

Sambalpur Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
raisen Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

raisen Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
nagpur Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

nagpur Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
❤️♀️@ Jaipur Call Girls ❤️♀️@ Jaispreet Call Girl Services in Jaipur QRYPCF ...

❤️♀️@ Jaipur Call Girls ❤️♀️@ Jaispreet Call Girl Services in Jaipur QRYPCF ...
Call Girls Patiala Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Patiala Just Call 9907093804 Top Class Call Girl Service Available
dhanbad Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

dhanbad Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
Call Now ☎ 9999965857 !! Call Girls in Hauz Khas Escort Service Delhi N.C.R.

Call Now ☎ 9999965857 !! Call Girls in Hauz Khas Escort Service Delhi N.C.R.
Call Girls Thane Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Thane Just Call 9907093804 Top Class Call Girl Service Available
Call Girls Hyderabad Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Hyderabad Just Call 9907093804 Top Class Call Girl Service Available
Russian Call Girls Kota * 8250192130 Service starts from just ₹9999 ✅

Russian Call Girls Kota * 8250192130 Service starts from just ₹9999 ✅
Call Girl Raipur 📲 9999965857 ヅ10k NiGhT Call Girls In Raipur

Call Girl Raipur 📲 9999965857 ヅ10k NiGhT Call Girls In Raipur
Ernakulam Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

Ernakulam Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
❤️Call girls in Jalandhar ☎️9876848877☎️ Call Girl service in Jalandhar☎️ Jal...

❤️Call girls in Jalandhar ☎️9876848877☎️ Call Girl service in Jalandhar☎️ Jal...
What Do Rheumatologists Think?
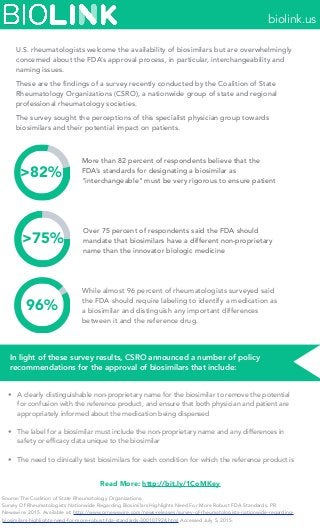
- 1. >82% >75% 96% U.S. rheumatologists welcome the availability of biosimilars but are overwhelmingly concerned about the FDA’s approval process, in particular, interchangeability and naming issues. These are the findings of a survey recently conducted by the Coalition of State Rheumatology Organizations (CSRO), a nationwide group of state and regional professional rheumatology societies. The survey sought the perceptions of this specialist physician group towards biosimilars and their potential impact on patients. More than 82 percent of respondents believe that the FDA’s standards for designating a biosimilar as "interchangeable" must be very rigorous to ensure patient Over 75 percent of respondents said the FDA should mandate that biosimilars have a different non-proprietary name than the innovator biologic medicine While almost 96 percent of rheumatologists surveyed said the FDA should require labeling to identify a medication as a biosimilar and distinguish any important differences between it and the reference drug. • A clearly distinguishable non-proprietary name for the biosimilar to remove the potential for confusion with the reference product, and ensure that both physician and patient are appropriately informed about the medication being dispensed • The label for a biosimilar must include the non-proprietary name and any differences in safety or efficacy data unique to the biosimilar • The need to clinically test biosimilars for each condition for which the reference product is In light of these survey results, CSRO announced a number of policy recommendations for the approval of biosimilars that include: Read More: http://bit.ly/1CoMKey Source:The Coalition of State Rheumatology Organizations. Survey Of Rheumatologists Nationwide Regarding Biosimilars Highlights Need For More Robust FDA Standards. PR Newswire. 2015. Available at: http://www.prnewswire.com/news-releases/survey-of-rheumatologists-nationwide-regarding- biosimilars-highlights-need-for-more-robust-fda-standards-300107924.html Accessed July 5, 2015. biolink.us
