sickle cell syndrome
- 2. CONTENTS: Introduction History Incidence Pathophysiology Types Genetic inheritence Clinical manifestations Investigations Treatment Complications Prognosis
- 3. INTRODUCTION Haemoglobinopathy refers to a disease state (opathy) involving the haemoglobin (Hb) molecule. Sickle cell syndrome is a type of qualitative haemoglobinopathy, occuring due to point mutation in β-like gene, leading to structural alteration in β-chain, of globin part of Hb and affecting the functional capacity of RBCs.
- 4. Sickle-cell disease (SCD), or sickle-cell anaemia or drepanocytosis, is an autosomal co-dominant genetic blood disorder characterized by red blood cell that assume an abnormal, rigid, sickle shape. +Recurrent episodes of painful crises, and progressive end-organ damage
- 5. HISTORY • Sickle cell anaemia was observed by a Chicago physician, James Herrick in 1910 in a West indian student with severe anaemia. • In 1927, Hahn and Gillespie described the pathologic basis of the disorder and its relationship to the haemoglobin molecule.
- 6. In 1946, Beet reported that malarial parasites were present less frequently in blood smears from patients with SCD compared with normal individuals. It was determined that sickle cell trait confers resistance against infection with plasmodium falciparum occuring early in childhood between the time, that passively acquired immunity passes and active immunity develops.
- 7. Pauling and Co-workers defined the genetics of the disorder and clearly distinguished heterozygous sickle trait (Hb AS)from the homozygous state (Hb SS).
- 8. INCIDENCE: The highest frequency of sickle cell trait is in Africa where the frequency is 20- 40%,the high prevalence can be explained by the selective survival advantage it confers in areas where Falciparum malaria is endemic. In the United states, approximately 12% of african Americans have a Haemoglobin variant.
- 9. It has also been found in individuals from the middle east, India, and the mediterranian. Also from regions of caribbean and central & South America. Sickle cell is becoming more prominent in southern India, particularly in certain tribes.
- 11. Incidence with Malaria The sickle gene occurs with greatest frequency in central africa ,the near East, the region around the mediterranian,and parts of India. The frequency of gene parallels the incidence of P. falciparum and seems to offer some protection in young patients with cerebral falciparum malaria.
- 14. Basic molecular lesion: In HbS, basic genetic defect is Single point mutation in one amino acid out of 146 in haemoglobin molecule. There is substitution of a Valine for Glutamic acid, at 6th residue position of β-Globin, producing Hb α₂ β₂ S Glutamic acid has a net charge of -1, whereas Valine as a net charge of +0. This amino acid substitution results in charge of +1, resulting a structural change in the Hb molecule.
- 15. Gower 1: 2 2 Gower 2: 2 2 Portland: 2 2 ------------------ F: 2 2 A2: 2 2 A: 2 2 < 2% < 3% 96% Hemoglobins by age > 1 yr
- 16. Molecular pathology of SCD The sickle mutation GAG Glutamic acid GTG Valine The s Mutation 6th Codon of -Globin Gene
- 17. In Sickle Cell Disease Gower 1: 2 2 Gower 2: 2 2 Portland: 2 2 ------------------ F: 2 2 A2: 2 2 S: 2 s 2 2-20% 3% 80-95% Hemoglobins in SS by age > 1 yr
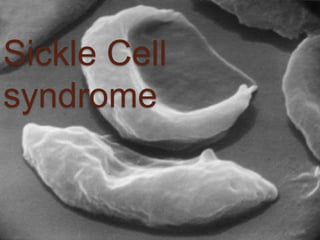
- 19. Mechanism of Sickling. During deoxygenation the red cells containing Hb S change from biconcave disc shape to an elongated Cresent shape, this process is ‘Sickling’. The Molecule of Hb under deoxygenated condition polymerize to form Pseudocrystalloid structures known as ‘Tactoids’. These aggregate to form elongated rod like polymers These elongated fibres align and distord the red cells to classic Sickle cell.
- 20. These polymerised deoxy-Hb S activates a membrane channel called Psickle that is otherwise inactive in normal RBCs. These membrane channels open when the blood Po2 decreases to less than 50mm Hg. Open Psickle channels allow the influx of Ca, raisin the intracellular Ca levels & activating a second membrane channel called the gardos channel. An activated gardos channel causes the efflux of K that stimulates the efflux of Cl through another membrane channel to maintain charge equilibrium across the RBC membrane.
- 21. The efflux of these ions leads to dehydration, effectively increasing the intracellular concentration of Hb S & intensifying polymerisation. OXY-STATE DEOXY-STATE
- 22. Reversible-Irreversible Sickling: The oxygen dependent process is usually reversible. However the damage to red cell membrane leads to the formation of irreversible sickled cell even after they are exposed to normal oxygen tension.
- 23. Factors determining the rate of Sickling: Presence of non-Hb S haemoglobins, Hb F. Intra cellular concentration of Hb S. Total haemoglobin concentrations. Extent of deoxygenation. Acidosis and Dehydration. Increased concentration of 2,3-BPG in the red cells.
- 24. Pathophysiology explaining ↑SCD in malarial endemic regions: Malarial parasite within RBC use the oxygen within the cell Reduce oxygen tension RBC Sickling, injury to cells Injured Cells get trapped in bleeding vessel
- 25. Easily phagocytosed by scavenger WBC’s. Selective destruction of Infected RBC’s containing Parasites. ↓↓ Number of Malarial organisms and ↑↑ in time for Immunity to develop.
- 26. TYPES: It occurs in 3 different forms: 1. Heterozygous state for Hb S: Sickle Cell Trait(AS) 2. Homozygous state for Hb S : Sickle Cell disease(SS) 3. Double heterozygous states eg: Sickle β-thalassemia Sickle C disease Sickle SC disease
- 27. Sickle Cell Trait Sickle haemoglobin (S) + Normal haemoglobin (A) in RBC • Adequate amount of normal Hb (A) in red blood cells • RBC remain flexible • Carrier • Do Not have the symptoms of the sickle cell disorders, • with exceptions: • Pain when Less Oxygen than usual (activities at high altitude (12,000ft), under general anaesthesia) • Minute kidney problems*
- 28. Haemoglobin C: Is the most common non sickling varient encountered Here lysine is substituted for glutamic acid in 6th position of β globin chain. Is inherited in same as HB S but manifest as a milder disease. Similar to Hb S, Hb C polymerises under low oxygen tension, but the structure of the polymers differs.
- 29. Hb polymers are long and thin whereas the polymers in Hb C form a short, thick crystal within the RBC. The shorter Hb C crystal does not alter RBC shape to the extent of Hb S, resulting in diminished splenic sequestration and hemolysis.
- 30. Hemoglobin SC Is the most common double heterozygous syndrome. At the 6th position, glutamic acid is replaced by valine(Hb S) on one β- globin chain & lysine(Hb C) on other β- globin chain. Hb SC disease resembles a mild SCD. Growth & development are delayed compared with normal children.
- 31. In contrast to SCD, significant symptoms here do not occur until teenage years. May cause all the vaso-occlusive complications of sickle cell anemia, but the episodes are less frequent, and damage is less debilitating.
- 32. Hemoglobin S/β-Thalassemia: Double heterozygosity for Hb S & β- thalassemia is the most common cause of sickle cell syndrome in patients of Mediterranean descent. It causes a clinical syndrome resembling that of mild or moderate sickle cell anaemia. The severity of this double heterozygous condition depends on β-chain production of the affected thalassemia β-genes.
- 33. If there is no globin chain production from the β-thalessaemia globin gene (S-β0- thal), the clinical course is similar to that of homozygous sickle cell anaemia If there is production 0f β globin (S-β+-thal), patients tend to have a milder condition than patients with Hb SC. These patients can be distinguished from individuals with Sickle cell trait -greater amount of Hb S than Hb A -↑↑ levels of Hb A2 and Hb F. -microcytosis from thalassaemia -hemolytic anaemia. -abnormal blood morphology. -splenomegaly.
- 34. GENETICS Sickle cell anemia is an autosomal co dominent genetic disorder. For the disease to be expressed, a person must inherit either two copies of Hb S variant(Sickle Cell Disease) or one copy of Hb S and one copy of another variant(Sickle Cell Trait)
- 35. Inheritance of Sickle Cell Anemia If both parents have sickle cell trait (HbAS) there is a one in four (25%) chance that any given child could be born with sickle cell anemia. There is also a one in four chance that any given child could be completely unaffected. There is a one in two (50%) chance that any given child will get the sickle cell trait
- 36. If one parent has sickle cell anaemia (HbSS) and the other is completely unaffected (HbAA) then all the children will have sickle cell trait. None will have sickle cell anemia. The parent who has sickle cell anemia (HbSS) can only pass the sickle hemoglobin gene to each of their children.
- 37. • If one parent has sickle cell trait (HbAS) and the other has sickle cell anaemia (HbSS) there is a one in two (50%) chance that any given child will get sickle cell trait and a one in two (50%) chance that any given child will get sickle cell anemia. • No children will be completely unaffected
- 38. • If one parent has sickle cell trait (HbAS) and the other does not carry the sickle hemoglobin at all (HbAA) then none of the children will have sickle cell anemia. • There is a one in two (50%) chance that any given child will get one copy of the HbAS gene and therefore have the sickle cell trait. • It is equally likely that any given child will get two HbAA genes and be completely unaffected.
- 41. The clinical manifestations of the sickle cell syndrome can vary from asymptomatic to a potentially lethal state as characterized by Sickle cell disease. Individuals affected with Sickle cell disease are characteristically asymptomatic until the the second half of the first year of life owing to the protective effect of Hb F.
- 42. During the first 6 months of life, mutated β chains are produced to gradually replace normal ᴕchains, causing Hb S levels to increase as Hb F levels decrease. Erythrocytes containing Hb S become susceptible to hemolysis, and a progressive hemolytic anaemia and slenomegaly may become evident.
- 43. Acute manifestations. Chronic manifestations. Vaso-occlusive crisis. -painful crisis. -acute chest syndrome. -Priapism. Cerebro-vascular events. -Thrombotic stroke. -Haemorrhagic stroke. Aplastic crisis. Splenic sequestration. Osteomyelitis. Chronic renal disease. -Isosthenuria. -CRF. Chronic pulmonary disease. Sickle hepatopathy. Proliferative Retinopathy. Avascular Necrosis. Skin ulcers.
- 44. Crisis: Typical course of sickle cell patient is, the period of relatively normal functioning, despite chronic anaemia, punctuated by periods of either pain in various anatomical sites, or other manifestations termed as “crisis” It is acute in onset These have been classified as -vaso occlusive painful crisis -aplastic crisis -sequestration crisis -hemolytic crisis
- 45. I. VASO-OCCLUSION Is the hallmark feature of SCD, accounting for most hospital & emergency dept visits. It results from complex interplay between sickled red cells, neutrophils, endothelium, & plasma factors. End result is that of tissue hypoxia leading to tissue death & accompanying pain. Fever is often present even in the absense of infection.
- 46. This acute painful aspect of SCD can be triggered by -acidosis -hypoxia -dehydration -infection -fever -extreme cold They manifest most often in the bones, lungs, liver, spleen, eyes, penis, CNS & urinary tract.
- 47. 1.BONES Pain The frequency of pain varies from none to six per year On average each episode persists for 4 to 5 days, although protracted episodes may last for weeks. Young children: mostly extremities Older patients: abdomen, back, chest Associated with intercurrent illness and other Vaso-occlusive event
- 48. Hand foot dactylitis. It’s a term used to describe painful swelling of digits of hands and feet(Hand- foot syndrome). Occurs early in infancy as Haematopoietic marrow is present in these bones at this
- 49. Most episodes resolve within 2 weeks. Epiphyseal infarction can result in joint pain and swelling mimicking septic arthritis.
- 50. Infection(osteomyelitis) . Impaired cellular and humoral immunity together with infarction of bone contribute to this condition. Non-typical serotypes of Salmonella, Staphylococcus, and Gram-Negative baccili are principle infection offenders.
- 51. 2. LUNGS Acute chest syndrome Is due to pulmonary infarction from sickling in microvasculature, infection or both. Also bone marrow infarction results in fat emboli to the lungs, being other cause. Is characterized by fever, acute chest pain & presence of pulmonary infiltrates on the Chest X ray. other manifestations:
- 52. In Children, acute chest syndrome generally is precipitated by infection characterized by Fever, cough, and tachypnea. In adults, this organ failure is the most common cause of Death.
- 53. 3.SPLEEN Sequestration splenomegaly Is usually seen under the age of 2 years, in infants and young children. The spleen rapidly enlarges (within hours) & most of the circulating red cell mass becomes sequestered. Is characterized by a sudden trapping of Blood in the spleen, resulting in rapid decline in Hb, often to less than 6g/dl, result in hypovolemic shock & cardiovascular collapse.
- 54. Characterised by rapid enlarging spleen, pain, hypoxemia and hypovolemic shock. These may be associated with fever, pain and respiratory symtoms. Circulatory collapse and death can occur in 30 min.
- 55. Auto splenectomy Multiple episodes of splenic sequestration leading to gradual loss of function(by the age of 6-8 yrs) termed as Auto- splenectomy . It confers a Non-functioning spleen, which is reduced to a shrunken, fibrosed structure. Hence patients are prone to microbial infections, especially encapsulated, such as -S. pneumoniae. -H. influenza
- 56. 4.LIVER Hepatomegaly. Chronic liver abnormality in SCD are frequent and are reflective of different etiologies of liver dysfunction that include Vaso-occlusion, Transfusion, Iron overload, Pigment gall stones, with bile duct obstruction, Acute or chronic cholecystitis, Viral Hepatitis and Cholestatsis.
- 57. Gallstones: Too much bilirubin from break down of Hb leads to gallstone formation. Manifest as Nausea, vomitting, fever, sweating , chills, clay coloured stools(Jaundice)
- 58. 5. Penis Priapism Is the painful erection of penis caused by sicklin red cells. This complication usually occurs in children and adolescents with SS or SC with an onset at age 5 to 35. It occurs as a severe episode requiring hospitalization following multiple episodes of short duration , termed stuttering. Onset in early morning, waking the patient is common. Impotence with severe disease or recurrent episodes Uret hra Corpus cavernosu
- 59. 6.EYES Retinal haemorrhage: Microvascular obstruction occurs followed by neovascularization and arterio venous aneurysm. Haemorrhage, scarring and retinal detachment leading to blindness are the sequel.
- 60. 7.CENTRAL NERVOUS SYSTEM: 25% of patients are affected with CNS manifestations. These include Transcient Ischaemic Attack, Strokes and Cerebral haemorrhage. Stroke: -in SCD is a macrovascular phenomenon, that affects approx. 11% of patients younger than 20 yrs. Is highest in first decade of life. Ischaemic stroke is most common in children & older adults, whereas haemorrhagic stroke in third decade of life.
- 61. Cerebral blood flow is significantly increased in SCD because of chronic anaemia & hypoxaemia. Cerebral vasculature is unable to vasodilate further in response to increased hypoxic stress thereby causing ischaemia. Risk factors: -Ischaemic stroke: TIA, recent or recurrent acute chest syndrome and Hypertention. -Haemorrrhagic stroke: Anaemia & Neutrophilia
- 62. 8.URINARY TRACT Renal failure: Incidence varies between 4-20% The acidic, hypoxic and hypertonic environment of the renal medulla promotes sickling of Hb SS RBCs leading to ischaemia of the renal microcirculation, loss of medullary function & renal papillary necrosis. Hematuria: Usually painless, can result from papillary necrosis, rupture of collateral vessels, & rarely from neoplasm.
- 63. Proteinuria/Nephrotic syndrome: 40% of SCD patients with nephrotic syndrome develop end-stage renal disease Occurs in ~ 20% of all patients Occurs in 4.5% of all pediatric patients - increased in Hemoglobin SS to 6.5% - Increased incidence with age. - Increased with anaemia, increased MCV, and increased leukocyte count.
- 64. 9.SKIN Leg ulcers It occurs in 2-40% cases of Sickle cell disease. Most common in patients older than 10 years of age. Typically ocurrs in lower extremities, especially on malleoli and cause chronic pain and disability. Venous stasis is the predisposing factor.
- 65. II. BACTERIAL INFECTIONS Sepsis. These patients have increased susceptibility to life threatening infections from S. aureus, St. pneumonia and H. influenza. Acute infections are common causes of hospitalizations and have been the most frequent cause of death, especially in the first 3 years of life.
- 66. These are exacerbated by auto- splenectomy effect as its ability to function as a secondary lymphoid tissue to clear organisms effectively from the blood is diminished, or lost. Pnemonia and Osteomyelitis.
- 67. III. HEMATOLOGICAL DEFECTS: Anaemia: Essentially a hemolytic process, can be correlated with intra-cellular polymer content. Characterised by generalised weakness, fatigue, shortness of breath on exertion, poor concentration.
- 68. Hemolytic crisis: Describes the occurnence of episodesof accelerated haemolysis characterised by decreased blood Hb, increased reticulocytes and other markers of haemolysis. Episodes of haemolysis occur in conditions like- -resolutive phase of Vaso-occlusive crisis. -delayed haemolytic transfusion reactions.
- 69. Megaloblastic episodes. This results from sudden arrest of erythropoiesis due to folate deficiency. FA requirement is higher in haemolytic anaemia and this factor is responsible for severe anaemia in patients who are nutritionally weak.
- 70. Aplastic crisis. Aplastic episodes (bone marrow failure) are the most common life threatening haemotological complications, and usually are associated with infection, particularly with Parvo viruses 19 resulting in a severe but self limiting red cell aplasia. SCD patients usual can compensate for the decrease in RBC survival by increasing bone marrow output.
- 71. But when bone marrow is suppressed temporarily by bacterial or viral infections however the haematocrit decreases substantially with no reticulocyte compensation. This produces a very low Hb which may cause heart failure.
- 72. IV. CARDIAC DEFECTS: Enlarged heart: the hemodynamic burden of anaemia results in elevated cardiac output and hence ventricular hypertrophy. Heart murmurs: -Forceful precordial apical impulse -Systolic & Diastolic flow murmers, as manifestation of hyperdynamic circulation.
- 73. 1. Fever 2. Chest pain 3. Shortness of Breath 4. Increasing tiredness 5. Abdominal swelling 6. Unusual Danger Signs of a Crisis 7. Any sudden weakness or loss of feeling 8. Pain that will not go away with home treatment . 9. Priapism (painful erection that will not go down) 10.Sudden vision change
- 74. V. OTHER FEATURES: Stunted growth: A vast majority of children with SCD will show decline in growth compared to normal peers. Puberty is delayed on an average by 12 to 24 months, as is skeletal age.
- 75. High risk pregnancy One third of patients will have adverse obstretic outcomes as defined by Miscarriages, StillBirths or Ectopic implantation. Preterm delivery occurs in 30-50% of pregnancy in sickle cell disease patients and 20% of infants have low birth weight.
- 76. A five fold increase in risk for Thromboembolism is present during pregnancy. Other significant findings include -higher rates of Caeserian section -infection -Gestational hypertension -Pre-eclampsia or Eclampsia -IUGR -asymptomatic bacteruria.
