Melden
Teilen
Downloaden Sie, um offline zu lesen
Empfohlen
Determination of potassium levels In 4 monitoring wells on Isle of Hope, Worm...

Determination of potassium levels In 4 monitoring wells on Isle of Hope, Worm...Fatai Olabanji Balogun
Stephanie Foltran (University of Nottingham) - Determination of Water Solubil...

Stephanie Foltran (University of Nottingham) - Determination of Water Solubil...UK Carbon Capture and Storage Research Centre
Determination of water solubility limits in CO2 mixtures to deliver water spe...

Determination of water solubility limits in CO2 mixtures to deliver water spe...UK Carbon Capture and Storage Research Centre
Activation of hydrogen peroxide by chemical reagent to reduce COD in petroche...

Activation of hydrogen peroxide by chemical reagent to reduce COD in petroche...Universiti Malaysia Pahang
Weitere ähnliche Inhalte
Was ist angesagt?
Determination of potassium levels In 4 monitoring wells on Isle of Hope, Worm...

Determination of potassium levels In 4 monitoring wells on Isle of Hope, Worm...Fatai Olabanji Balogun
Stephanie Foltran (University of Nottingham) - Determination of Water Solubil...

Stephanie Foltran (University of Nottingham) - Determination of Water Solubil...UK Carbon Capture and Storage Research Centre
Determination of water solubility limits in CO2 mixtures to deliver water spe...

Determination of water solubility limits in CO2 mixtures to deliver water spe...UK Carbon Capture and Storage Research Centre
Was ist angesagt? (13)
Quantification of Copper in Surface Sediment Samples via Flame Atomic Absorpt...

Quantification of Copper in Surface Sediment Samples via Flame Atomic Absorpt...
Determination of potassium levels In 4 monitoring wells on Isle of Hope, Worm...

Determination of potassium levels In 4 monitoring wells on Isle of Hope, Worm...
Stephanie Foltran (University of Nottingham) - Determination of Water Solubil...

Stephanie Foltran (University of Nottingham) - Determination of Water Solubil...
Integrated Electrodialysis Membrane Stability Results for Cost-Effective CBM ...

Integrated Electrodialysis Membrane Stability Results for Cost-Effective CBM ...
C:\Documents And Settings\Jbalent\My Documents\Tio061610

C:\Documents And Settings\Jbalent\My Documents\Tio061610
Determination of water solubility limits in CO2 mixtures to deliver water spe...

Determination of water solubility limits in CO2 mixtures to deliver water spe...
Ähnlich wie Hw3 with answer
Activation of hydrogen peroxide by chemical reagent to reduce COD in petroche...

Activation of hydrogen peroxide by chemical reagent to reduce COD in petroche...Universiti Malaysia Pahang
Ähnlich wie Hw3 with answer (20)
0.0 EXPERIMENT ON DETERMINATION OF DISSOLVED OXYGEN

0.0 EXPERIMENT ON DETERMINATION OF DISSOLVED OXYGEN
Activation of hydrogen peroxide by chemical reagent to reduce cod in petroche...

Activation of hydrogen peroxide by chemical reagent to reduce cod in petroche...
Activation of hydrogen peroxide by chemical reagent to reduce COD in petroche...

Activation of hydrogen peroxide by chemical reagent to reduce COD in petroche...
Laboratory manual of water supply and sewerage engineering

Laboratory manual of water supply and sewerage engineering
Biological Oxygen Demand Process and Explanation.pptx

Biological Oxygen Demand Process and Explanation.pptx
Mehr von Richard Templin
Mehr von Richard Templin (14)
Hw3 with answer
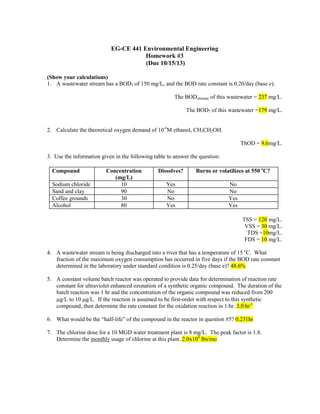
- 1. EG-CE 441 Environmental Engineering Homework #3 (Due 10/15/13) (Show your calculations) 1. A wastewater stream has a BOD5 of 150 mg/L, and the BOD rate constant is 0.20/day (base e). The BODultimate of this wastewater = 237 mg/L. The BOD7 of this wastewater =179 mg/L. 2. Calculate the theoretical oxygen demand of 10-4M ethanol, CH3CH2OH. ThOD = 9.6mg/L. 3. Use the information given in the following table to answer the question: Compound Sodium chloride Sand and clay Coffee grounds Alcohol Concentration (mg/L) 10 90 30 80 Dissolves? Burns or volatilizes at 550 oC? Yes No No Yes No No Yes Yes TSS = 120 mg/L. VSS = 30 mg/L. TDS =10mg/L. FDS = 10 mg/L. 4. A wastewater stream is being discharged into a river that has a temperature of 15 oC. What fraction of the maximum oxygen consumption has occurred in five days if the BOD rate constant determined in the laboratory under standard condition is 0.25/day (base e)? 48.6% 5. A constant volume batch reactor was operated to provide data for determination of reaction rate constant for ultraviolet enhanced ozonation of a synthetic organic compound. The duration of the batch reaction was 1 hr and the concentration of the organic compound was reduced from 200 µg/L to 10 µg/L. If the reaction is assumed to be first-order with respect to this synthetic compound, then determine the rate constant for the oxidation reaction in 1/hr. 3.0 hr-1 6. What would be the “half-life” of the compound in the reactor in question #5? 0.231hr 7. The chlorine dose for a 10 MGD water treatment plant is 8 mg/L. The peak factor is 1.8. Determine the monthly usage of chlorine at this plant. 2.0x104 lbs/mo
