Session 4 part 5
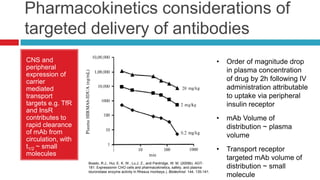
- 1. Pharmacokinetics considerations of targeted delivery of antibodies CNS and • Order of magnitude drop peripheral in plasma concentration expression of carrier of drug by 2h following IV mediated administration attributable transport to uptake via peripheral targets e.g. TfR insulin receptor and InsR contributes to • mAb Volume of rapid clearance distribution ~ plasma of mAb from volume circulation, with t1/2 ~ small • Transport receptor molecules targeted mAb volume of Boado, R.J., Hui, E. K. W., Lu,J. Z., and Pardridge, W. M. (2009b). AGT- 181: Expressionin CHO cells and pharmacokinetics, safety, and plasma distribution ~ small iduronidase enzyme activity in Rhesus monkeys.). Biotechnol. 144, 135-141. molecule
- 2. Challenges Associated with Targeted Delivery for CNS indications Advantages of Traditional mAbs Bi-specific targeting modalities, e.g. Long t1/2 BACE IV-transfusion, infrequent dosing Scalable manufacture of bi-specific (monthly) mAb PK Advantages Negated by transport Cost of Goods: receptor targeted delivery Hu eq dose BACE/TfR = 1.75g/70kg; More Frequent dosing depending Tysabri: 300 mg IV, q4 wks upon: Humira: 40-160 mg IV, qw – q4 wk Target:Ligand stoichiometry demands Dosing interval BACE/TfR? for desired pharmacologic outcome Monthly = 21g/person/yr Pharmacodynamic effect if target engagement may allow less frequent Bimonthly = 42g/person/yr dosing 300 person 1 yr P2 trial = 12.6 kg drug product
- 3. Antibody Technologies CMC, timeline to IND, and cost considerations
- 4. Growth of Antibody Therapeutics Nelson AL, Dhimolea E, Reichert JM. 2010. Development trends for human monoclonal antibody therapeutics. Nature reviews Drug discovery 9: 767-774.
- 5. Upfront considerations with antibody therapeutics Advantages: Excellent target selectivity (safety), Ka Predictable PK, metabolism, and elimination Cross reactive with antigen in humans and in preclinical model Straight forward path to clinical development with pre-clinically active agent Humanized or Human mAb via appropriate technology Neutralizing agent in preclinical model does not recognize Hu antigen, or with less activity (e.g. Ka) Ab discovery to identify lead with equipotent activity against Hu Ag Demonstrate bio-equivalence of anti-Hu mAb with pre-clinically active lead Confirm activity of hu mAb in pre-clinical model of disease Primates Rodents Acute (target engagement) vs chronic (treatment) paradigm Induce tolerance to huAb in rodent model
- 6. Antibody Technologies Mouse – Human • CDR Grafted • Humanized Fully Human • Hu Hybridomas • Phage Display • Tg Mouse • Direct cloning from Brekke, O. H. and I. Sandlie (2003). "Therapeutic antibodies for human diseases at the dawn of Patient/Dono the twenty-first century." Nat Rev Drug Discov 2(1): 52-62. r B-cells
- 7. Antibody Humanization Humanized Mouse Hybridoma VL + VH VH-C 1 rDNA VL-C VL C Sequence informatics VH C 1 + Molecular Modeling Transfected Cell VH-C 1 VL-C Murine mAb Chimeric mAb
- 8. CM&C: Biologics v small molecules Terminology, jargon, and lexicon Upstream: scale up production of composition of matter/drug substance Downstream: Purification, formulation, stability of drug product Drug substance – Comp of matter, pre formulation to post formulation (i.e. incl. excipients stability, buffers etc.) bulk substance Drug Product – Final formulation of DP in labeled vial, pills, or capsules, as it will be administered to patients Sm Molecule – customized mfg for ea candidate dictated by composition of matter Biologic – Platform process, upstream & downstream Composition of matter – rDNA technology, vectors Cell based production, scalable bioreactor technology + process improvements = Titers≥1g/L Downstream Extensive list of QA/QC tests for DP
- 9. Manufacturing timeline and $ for typical mAb („whaddya mean no IND this year?) Months 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 Stage Cell Line Dev. PD & Tox GMP (Clinic) Material Supplied RCB MCB 500g Tox 1.5Kg Clinic Vector Generation Cell Line Development CLD: $1.5M Upstream Development Downstream Development Formulation and Analytical Dev DS Mfg: $6.2M MCB and WCB Generation Tech Xfer / Tox / GMP runs Fill / Analysis /Release DP Mfg: $0.8M
- 10. “Caveat Entrepreneur(?)” “The first one out the door gets all the arrows in his back” * *Michael West, Geron CEO, Nature 479: 459 Steinmetz & Spack, BMC Neurology 2009, 9(Suppl 1):S2 doi:10.1186/1471-2377-9-S1-S2
- 11. Summary Opportunities for biologics in Neurodegenerative disease Apply & Exercise Sound Principles of Drug Discovery Targeted delivery poses challenges from perspective of drug development Antibody therapeutics offer robust, scalable platform with proven market value, at a cost
Hinweis der Redaktion
- Google success rates of mAbsvs Small molecules
- Apredica Poster
- Accompany slide with PR of JNJ/Pfizer mergers