2 4 What Is Pressure
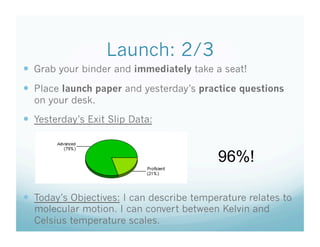
- 1. Launch: 2/3 Grab your binder and immediately take a seat! Place launch paper and yesterday’s practice questions on your desk. Yesterday’s Exit Slip Data: 96%! Today’s Objectives: I can describe temperature relates to molecular motion. I can convert between Kelvin and Celsius temperature scales.
- 2. Launch: 2/3 Grab your binder and immediately take a seat! Place launch paper and yesterday’s practice questions on your desk. Yesterday’s Exit Slip Data: 95%! Today’s Objectives: I can describe temperature relates to molecular motion. I can convert between Kelvin and Celsius temperature scales.
- 3. Launch: 2/3 Grab your binder and immediately take a seat! Place launch paper and yesterday’s practice questions on your desk. Yesterday’s Exit Slip Data: 97% Today’s Objectives: I can describe temperature relates to molecular motion. I can convert between Kelvin and Celsius temperature scales.
- 4. Launch: 2/3 Grab your binder and immediately take a seat! Place launch paper and yesterday’s practice questions on your desk. Yesterday’s Exit Slip Data: 95% Today’s Objectives: I can describe how temperature relates to molecular motion. I can convert between Kelvin and Celsius temperature scales.
- 5. Launch 2/3 1. Draw a picture of perfume from a beautiful lady (or cologne from a gorgeous man) diffusing across a dance floor to your nose. • How do the gas molecules move?
- 6. Launch 2/3 2. After one gas fully diffuses into another, the gas molecules are a. slowed down by the increase in ionic interactions. b. considered to be a heterogeneous mixture. c. not as likely to react chemically. d. evenly mixed together and evenly distributed around their container.
- 7. Launch 2/3 3. If gas molecules move really fast, why does it take a few minutes for someone to smell perfume across a large room? a. the increase in the airspace occupied by perfume molecules b. the chemical reaction with the nerves, with is slower than other sensory processes c. intermolecular forces between the air and perfume molecules d. random collisions between the air and perfume molecules
- 8. Announcements Unit #7: Gases New unit Start of on the right foot Quiz on Friday!
- 9. Monday February Calendar LPS Chem, Assessments, Unit Plan Tuesday Wednesday Thursday Feb 2010 (Pacific Time) Friday 1 2 3 4 5 Staff PD No School Gases and Their Properties Today Quiz! 8 9 10 11 12 Gases and Their Properties Quiz! 15 16 17 18 19 President's Week No School 22 23 24 25 26 Gases and Their Properties Exam!
- 10. Opening Today’s Objective: I can describe how the random collisions between gas molecules creates pressure on a surface. Standard – CH.4.a Today’s Questions: What is pressure?
- 11. What is pressure? Mr. Heffner 2/4/10
- 12. What is pressure? Pressure is… a measure of the force that gases exert on an object This force comes from… the random collisions of gas molecules with the walls of a container
- 13. What is pressure? Pressure increases when… the # of collisions increases What the speed of the gas molecules increases increases the speed? …temperature is increased! Molecular Motion Applet
- 14. What is pressure? temperature = collisions = pressure
- 15. What is pressure? Pressure is measured in units of… 1. atmospheres (atm) 1 atm = pressure @ sea level 2. millimeters of mercury (mmHg) 760 mmHg = pressure @ sea level 1 atm = 760 mmHg
- 16. Foldable Define the following terms: 1. pressure 2. temperature
- 17. Practice Questions Practice question worksheet
- 18. Closing Today’s Objective: I can describe how temperature relates to molecular motion. I can convert between Kelvin and Celsius temperature scales. Standard – CH.4.e & CH.4.a Molecular Motion Applet
- 19. Exit Slip 1. Temperature measures a. the height at which gas molecules travel in the atmosphere b. the amount of kinetic energy in a substance c. quantities in units of °G d. the time it takes for the weather to change
- 20. Exit Slip 2. Molecules in a substance at 50°C a. move faster than molecules in a substance at 25°C because they have less kinetic energy b. move faster than molecules in a substance at 25°C because they have more kinetic energy c. move slower than molecules in a substance at 25°C because they have less kinetic energy d. move slower than molecules in a substance at 25°C because they have more kinetic energy
- 21. Exit Slip 3. Which of the following is the correct equation for converting between Kelvin and Celsius temperature scales? a. K = °C + 273 b. °C = K + 273 c. K = C + 373 d. °C = K + 373
- 22. Exit Slip 4. Water boils at 100°C. What is that temperature in Kelvin? a. -173K b. 100K c. 173K d. 373K
- 23. Exit Slip 5. What is the equivalent of 423K in degrees Celsius? a. -223°C b. -23°C c. 150°C d. 696°C
- 24. Homework Finish practice questions