1 14 What Is Concentration Part Iii
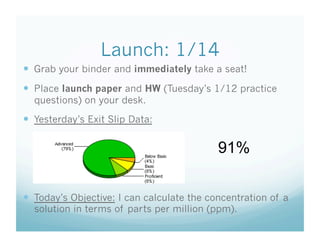
- 1. Launch: 1/14 Grab your binder and immediately take a seat! Place launch paper and HW (Tuesday’s 1/12 practice questions) on your desk. Yesterday’s Exit Slip Data: 91% Today’s Objective: I can calculate the concentration of a solution in terms of parts per million (ppm).
- 2. Launch: 1/14 Grab your binder and immediately take a seat! Place launch paper and HW (Tuesday’s 1/12 practice questions) on your desk. Yesterday’s Exit Slip Data: 87% Today’s Objective: I can calculate the concentration of a solution in terms of parts per million (ppm).
- 3. Launch: 1/14 Grab your binder and immediately take a seat! Place launch paper and HW (Tuesday’s 1/12 practice questions) on your desk. Yesterday’s Exit Slip Data: 90% Today’s Objective: I can calculate the concentration of a solution in terms of parts per million (ppm).
- 4. Launch: 1/14 Grab your binder and immediately take a seat! Place launch paper and HW (Tuesday’s 1/12 practice questions) on your desk. Yesterday’s Exit Slip Data: 90% Today’s Objective: I can calculate the concentration of a solution in terms of parts per million (ppm).
- 5. Launch 1/14 1. Concentration is a. a way to describe the motion of solvent particles in a solution. b. a measure of how quickly a solute dissolves. c. a number that can be measured in g/L or mol/L. d. how well a solute dissolves in a given amount of solvent.
- 6. Launch 1/14 3. If 18 mol of NaCl is dissolved in 9L of water, what is the molarity of the solution in mol/L? a. .5 mol/L b. 2 mol/L c. 3 mol/L d. 162 mol/L
- 7. Launch 1/14 4. What is the molarity of a solution that has 20 moles of HNO3 dissolved into 5L of water. a. 2 M b. 4 M c. 20 M d. 100 M
- 8. Launch 1/14 5. How many moles of NaOH are needed to make 10L of a 5M solution? a. .5 mol b. 2 mol c. 25 mol d. 50 mol
- 9. Announcements The semester ends next Friday Quiz tomorrow – 25 questions Take home notes/practice questions and study hard! Look at exit slips online! Quiz next Friday
- 10. January Calendar
- 11. What is concentration? – Part III Mr. Heffner 1/14/10
- 12. What is concentration? Concentration is… the amount of solute dissolved a in solution. Concentration is measured in… 1. grams per liter (g/L) 2. moles per liter (mol/L)
- 13. What is concentration? Concentration is also measured in… 3. parts per million (ppm) the # of solute particles dissolved in 1,000,000 solvent particles # solute particles 1,000,000 solvent particles
- 14. Examples 1. A solution of salt water contains 15 grams of salt per 1,000 grams of water. What is the concentration in parts per million (ppm)? 15 ? 1000 = 1000000 15 15000 1000 = 1000000 15,000ppm
- 15. Examples 2. A solution of sugar water contains 300 grams of sugar per 10,000 grams of water. What is the concentration in parts per million (ppm)? 300 ? 10000 = 1000000 300 30000 10000 = 1000000 30,000ppm
- 16. Examples 3. What is the concentration in parts per million (ppm) of a solution of salt water that contains 2 grams of salt per 100,000 grams of water? 2 ? 100000 = 1000000 2 20 100000 = 1000000 20ppm
- 17. Examples 4. A solution of salt water contains 395 grams of salt per 1000 grams of water. What is the concentration in parts per million (ppm)? 395 ? 1000 = 1000000 395 395000 1000 = 1000000 395,000ppm
- 18. Review: Solutes vs. Solvent A solution is a homogeneous mixture Solute = is dissolved, a solid (or gas), doesn’t move Ex: salt Solvent = does the dissolving, a liquid, moves randomly Ex: water (polar solvent)
- 19. Review: How are solutes dissolved? In a solution… 1. The solute (solid) is attacked by the solvent (liquid) 2. Solid-solid attractions are overpowered by the liquid 3. The solute is surrounded = dissolved
- 20. Review: What is solubility? Factors that affect solubility of solids 1. Temperature: temperature = solubility 2. Surface Area: surface area = solubility more SA less SA Factors that affect solubility of gases 1. Temperature: temperature = solubility 2. Pressure: pressure = solubility
- 21. Review: Concentration Calculations If the question asks for: g/L or mol/L or molarity divide grams or moles multiply ppm ratio M = molarity = mol/L
- 22. Solutions Rotations Directions: Pick a question and write it down Solve showing all work When finished, Mr. Heffner will come around to stamp your work Rules: If you pick it, solve it Only one card at a time Show your work! Work together
- 23. Homework Study for the quiz!