Ch 17 acid base
•Als PPTX, PDF herunterladen•
1 gefällt mir•22,995 views
Melden
Teilen
Melden
Teilen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
chemistry ch 2-acids,bases and salts by shamil shanavas

chemistry ch 2-acids,bases and salts by shamil shanavas
Chapter - 5, Acids, Bases and Salts, Science, Class 7

Chapter - 5, Acids, Bases and Salts, Science, Class 7
Andere mochten auch
Andere mochten auch (20)
Planetarymotionhistoricalpeopleinastronomy 131204162554-phpapp02

Planetarymotionhistoricalpeopleinastronomy 131204162554-phpapp02
Kasus Kecil Interna : CKD, Hipertensi, Diabetes Melitus, CHF

Kasus Kecil Interna : CKD, Hipertensi, Diabetes Melitus, CHF
Ähnlich wie Ch 17 acid base
Ähnlich wie Ch 17 acid base (20)
Kürzlich hochgeladen
No-1 Call Girls In Goa 93193 VIP 73153 Escort service In North Goa Panaji, Ca...

No-1 Call Girls In Goa 93193 VIP 73153 Escort service In North Goa Panaji, Ca...Call girls in Goa, +91 9319373153 Escort Service in North Goa
Call Us ➥9319373153▻Call Girls In North Goa

Call Us ➥9319373153▻Call Girls In North GoaCall girls in Goa, +91 9319373153 Escort Service in North Goa
Enjoy ➥8448380779▻ Call Girls In Sector 18 Noida Escorts Delhi NCR

Enjoy ➥8448380779▻ Call Girls In Sector 18 Noida Escorts Delhi NCRStunning ➥8448380779▻ Call Girls In Hauz Khas Delhi NCR
Kürzlich hochgeladen (20)
Ms Motilal Padampat Sugar Mills vs. State of Uttar Pradesh & Ors. - A Milesto...

Ms Motilal Padampat Sugar Mills vs. State of Uttar Pradesh & Ors. - A Milesto...
No-1 Call Girls In Goa 93193 VIP 73153 Escort service In North Goa Panaji, Ca...

No-1 Call Girls In Goa 93193 VIP 73153 Escort service In North Goa Panaji, Ca...
Islamabad Escorts | Call 03070433345 | Escort Service in Islamabad

Islamabad Escorts | Call 03070433345 | Escort Service in Islamabad
8447779800, Low rate Call girls in Shivaji Enclave Delhi NCR

8447779800, Low rate Call girls in Shivaji Enclave Delhi NCR
MAHA Global and IPR: Do Actions Speak Louder Than Words?

MAHA Global and IPR: Do Actions Speak Louder Than Words?
Call Girls in DELHI Cantt, ( Call Me )-8377877756-Female Escort- In Delhi / Ncr

Call Girls in DELHI Cantt, ( Call Me )-8377877756-Female Escort- In Delhi / Ncr
NewBase 19 April 2024 Energy News issue - 1717 by Khaled Al Awadi.pdf

NewBase 19 April 2024 Energy News issue - 1717 by Khaled Al Awadi.pdf
Call US-88OO1O2216 Call Girls In Mahipalpur Female Escort Service

Call US-88OO1O2216 Call Girls In Mahipalpur Female Escort Service
Investment in The Coconut Industry by Nancy Cheruiyot

Investment in The Coconut Industry by Nancy Cheruiyot
Memorándum de Entendimiento (MoU) entre Codelco y SQM

Memorándum de Entendimiento (MoU) entre Codelco y SQM
8447779800, Low rate Call girls in Kotla Mubarakpur Delhi NCR

8447779800, Low rate Call girls in Kotla Mubarakpur Delhi NCR
Enjoy ➥8448380779▻ Call Girls In Sector 18 Noida Escorts Delhi NCR

Enjoy ➥8448380779▻ Call Girls In Sector 18 Noida Escorts Delhi NCR
Ch 17 acid base
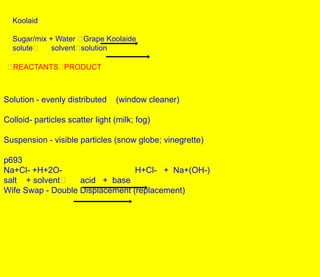
- 1. Koolaid Sugar/mix + Water Grape Koolaide solute solvent solution REACTANTS PRODUCT Solution - evenly distributed (window cleaner) Colloid- particles scatter light (milk; fog) Suspension - visible particles (snow globe; vinegrette) p693 Na+Cl- +H+2O- H+Cl- + Na+(OH-) salt + solvent acid + base Wife Swap - Double Displacement (replacement)
- 2. Concentration Dilute - little solute Concentrated - lot solute Solubitity - how much solute can be dissolved by a certain amount of solvent at a certain temperature saturated can not add anymore unsaturated can add more supersaturated over the amount (rock candy) Affect solubility- temperature pressure type of solvent ( Nail water/acid)
- 3. Acids Bases H+(hydronium) OH- (Hydroxide ion) sour bitter red litmus blue litmus dissolve metals slippery Oh! The bitter blue bases are slippery. Indicatiors litmus paper (blue/red) pH paper (colors) red cabbage juice (red.pink.purple. greens) phenolphthalein (colorless turn base pink) pH Scale 0-14 1-6 acid 7 neutral 8-14 bases
- 4. 1 strong acid - 6 wk acid 7 neutral 8 wk base - 14 strong base 1 stomach acid 2 lemon 3 grapes 4 tomato 5 bananas 6 milk- Laectic Acid 7 water 8 potato 9 baking soda 10 soap 12 Ammonia 14 Lye
- 5. Neutralization acid + base salt + Water H+Cl- + Na+OH- Na+ Cl- + H2O Double Displacement (Replacement) (wife swap)
- 6. Digestion Mouth Mechanical Chewing salivia pH neutral Stomach Mechanical/ Chemical acid pH 2(hydrocholoric acid) Intestine Mechanical/Chemical base pH 8(bicarbonate ions) Digestive enzymes work better at different pH's. enzymes are catalysts. carbohydrates proteins fats