Thermo#1
•Als PPTX, PDF herunterladen•
0 gefällt mir•331 views
Melden
Teilen
Melden
Teilen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (19)
Wk 5 p1 wk 6-p2_12.1-12.2_thermal properties of materials

Wk 5 p1 wk 6-p2_12.1-12.2_thermal properties of materials
Andere mochten auch
Andere mochten auch (7)
Ähnlich wie Thermo#1
Ähnlich wie Thermo#1 (20)
20- Explain the difference between temperature and heat- Also- state w.docx

20- Explain the difference between temperature and heat- Also- state w.docx
Diploma_I_Applied science(chemistry)U-V Thermodynamics 

Diploma_I_Applied science(chemistry)U-V Thermodynamics
Mehr von gbsliebs2002
Mehr von gbsliebs2002 (20)
Kürzlich hochgeladen
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...?#DUbAI#??##{{(☎️+971_581248768%)**%*]'#abortion pills for sale in dubai@
Kürzlich hochgeladen (20)
Apidays Singapore 2024 - Building Digital Trust in a Digital Economy by Veron...

Apidays Singapore 2024 - Building Digital Trust in a Digital Economy by Veron...
How to Troubleshoot Apps for the Modern Connected Worker

How to Troubleshoot Apps for the Modern Connected Worker
Apidays New York 2024 - The value of a flexible API Management solution for O...

Apidays New York 2024 - The value of a flexible API Management solution for O...
TrustArc Webinar - Stay Ahead of US State Data Privacy Law Developments

TrustArc Webinar - Stay Ahead of US State Data Privacy Law Developments
Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...

Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...
Polkadot JAM Slides - Token2049 - By Dr. Gavin Wood

Polkadot JAM Slides - Token2049 - By Dr. Gavin Wood
Boost Fertility New Invention Ups Success Rates.pdf

Boost Fertility New Invention Ups Success Rates.pdf
Powerful Google developer tools for immediate impact! (2023-24 C)

Powerful Google developer tools for immediate impact! (2023-24 C)
Strategize a Smooth Tenant-to-tenant Migration and Copilot Takeoff

Strategize a Smooth Tenant-to-tenant Migration and Copilot Takeoff
Apidays New York 2024 - The Good, the Bad and the Governed by David O'Neill, ...

Apidays New York 2024 - The Good, the Bad and the Governed by David O'Neill, ...
Automating Google Workspace (GWS) & more with Apps Script

Automating Google Workspace (GWS) & more with Apps Script
2024: Domino Containers - The Next Step. News from the Domino Container commu...

2024: Domino Containers - The Next Step. News from the Domino Container commu...
"I see eyes in my soup": How Delivery Hero implemented the safety system for ...

"I see eyes in my soup": How Delivery Hero implemented the safety system for ...
Thermo#1
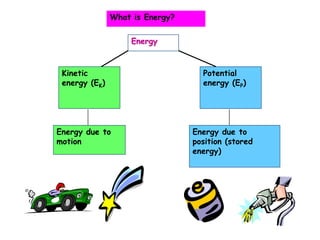
- 1. Energy Kinetic energy (EK) Potential energy (EP) Energy due to motion Energy due to position (stored energy) What is Energy?
- 2. Total Energy = Kinetic Energy + Potential Energy E = EK + EP Kinetic energy & potential energy are interchangeable Ball thrown upwards slows & loses kinetic energy but gains potential energy The reverse happens as it falls back to the ground
- 3. Law of Conservation of Energy: the total energy of the universe is constant and can neither be created nor destroyed; it can only be transformed. The internal energy, U, of a sample is the sum of all the kinetic and potential energies of all the atoms and molecules in a sample i.e. it is the total energy of all the atoms and molecules in a sample
- 4. State Functions A property of a system that changes independently of its pathway Energy changes this way
- 5. Temperature vs. Heat Temperature (T) is a measure of the kinetic energy of particles’ random motion (°C, °F or K) Heat (q) is a measure of the total amount of energy transferred from an object of high temperature to one of low temperature (J or cal)
- 7. The Ultimate Formula (not really) The heat energy needed to raise a substance to a certain temperature is related to the mass of the substance and the temperature change (ΔT) q = mass x specific heat x temperature change q = m CpΔT