Electronegativity Difference
•Als PPT, PDF herunterladen•
1 gefällt mir•379 views
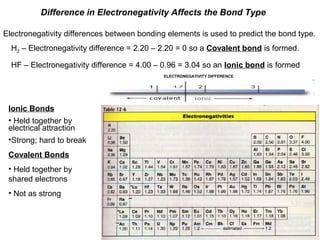
Chart depicting the electronegativity differences of atoms, including definitions and examples of ionic and covalent bonding in the context of electronegativity.
Melden
Teilen
Melden
Teilen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Mehr von Emma Wise
Mehr von Emma Wise (20)
Cleveland's Got It All CD and the Greater Cleveland Food Bank 

Cleveland's Got It All CD and the Greater Cleveland Food Bank
How to Help the Humane Society of Central Illinois

How to Help the Humane Society of Central Illinois
Kürzlich hochgeladen
Faculty Profile prashantha K EEE dept Sri Sairam college of Engineering

Faculty Profile prashantha K EEE dept Sri Sairam college of EngineeringSri Sairam College Of Engineering Bengaluru
31 ĐỀ THI THỬ VÀO LỚP 10 - TIẾNG ANH - FORM MỚI 2025 - 40 CÂU HỎI - BÙI VĂN V...

31 ĐỀ THI THỬ VÀO LỚP 10 - TIẾNG ANH - FORM MỚI 2025 - 40 CÂU HỎI - BÙI VĂN V...Nguyen Thanh Tu Collection
Mattingly "AI & Prompt Design: Large Language Models"

Mattingly "AI & Prompt Design: Large Language Models"National Information Standards Organization (NISO)
Kürzlich hochgeladen (20)
4.16.24 21st Century Movements for Black Lives.pptx

4.16.24 21st Century Movements for Black Lives.pptx
Visit to a blind student's school🧑🦯🧑🦯(community medicine)

Visit to a blind student's school🧑🦯🧑🦯(community medicine)
Mental Health Awareness - a toolkit for supporting young minds

Mental Health Awareness - a toolkit for supporting young minds
Oppenheimer Film Discussion for Philosophy and Film

Oppenheimer Film Discussion for Philosophy and Film
MS4 level being good citizen -imperative- (1) (1).pdf

MS4 level being good citizen -imperative- (1) (1).pdf
prashanth updated resume 2024 for Teaching Profession

prashanth updated resume 2024 for Teaching Profession
Transaction Management in Database Management System

Transaction Management in Database Management System
Faculty Profile prashantha K EEE dept Sri Sairam college of Engineering

Faculty Profile prashantha K EEE dept Sri Sairam college of Engineering
Student Profile Sample - We help schools to connect the data they have, with ...

Student Profile Sample - We help schools to connect the data they have, with ...
Q-Factor General Quiz-7th April 2024, Quiz Club NITW

Q-Factor General Quiz-7th April 2024, Quiz Club NITW
31 ĐỀ THI THỬ VÀO LỚP 10 - TIẾNG ANH - FORM MỚI 2025 - 40 CÂU HỎI - BÙI VĂN V...

31 ĐỀ THI THỬ VÀO LỚP 10 - TIẾNG ANH - FORM MỚI 2025 - 40 CÂU HỎI - BÙI VĂN V...
Mattingly "AI & Prompt Design: Large Language Models"

Mattingly "AI & Prompt Design: Large Language Models"
Electronegativity Difference
- 1. Difference in Electronegativity Affects the Bond Type Electronegativity differences between bonding elements is used to predict the bond type. H2 – Electronegativity difference = 2.20 – 2.20 = 0 so a Covalent bond is formed. HF – Electronegativity difference = 4.00 – 0.96 = 3.04 so an Ionic bond is formed ELECTRONEGATIVITY DIFFERENCE Ionic Bonds • Held together by electrical attraction •Strong; hard to break Covalent Bonds • Held together by shared electrons • Not as strong
