Axoplasmic flow in Axons - Mechanisms and Applications in Clinical Neurology
- 2. Road Map for the Session • • • • • • • Introduction and need for this discussion Historical aspects and the pioneers Characterization of the types of axoplasmic flow The molecular “motors” Integration of concepts Clinical utilization of the information – pathogenesis Clinical utilization of the information – therapeutics • Further reading
- 3. Road Map for the Session • • • • • • • Introduction and need for this discussion Historical aspects and the pioneers Characterization of the types of axoplasmic flow The molecular “motors” Integration of concepts Clinical utilization of the information – pathogenesis Clinical utilization of the information – therapeutics • Further reading
- 5. The Neuron • Largest and longest cells of body (107 rbc)
- 6. All proteins have to come from the soma
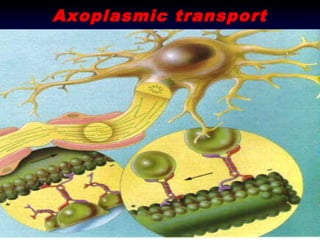
- 7. “Axoplasmic flow” • Conveyor Belt • Passive Transport • Gravity dependent
- 8. Road Map for the Session • • • • • • • Introduction and need for this discussion Historical aspects and the pioneers Characterization of the types of axoplasmic flow The molecular “motors” Integration of concepts Clinical utilization of the information – pathogenesis Clinical utilization of the information – therapeutics • Further reading
- 9. Pioneering Work • Concept of Axoplasmic Flow • Simple, elegant experiment • Rat Sciatic Nerve
- 11. Initial Response Jordi Floch, Founder AAN – “Thank God! What do you think the nervous system is, a plumbing system ?”
- 12. Road Map for the Session • • • • • • • Introduction and need for this discussion Historical aspects and the pioneers Characterization of the types of axoplasmic flow The molecular “motors” Integration of concepts Clinical utilization of the information – pathogenesis Clinical utilization of the information – therapeutics • Further reading
- 13. Advent of electron microscopy • Late 1960’s • Characterization of Sub cellular structure of Neuron • Absence of Golgi apparatus, RER and centromere from Axon • Presence of cytoskeletal proteins, vesicles, neurofilaments and neurotubules in axon
- 20. Classification of Axonal Flow • Slow Transport – Antegrade, 0.1 to 4 mm/day
- 25. Requires ATP/Mg2+ as fuel for the motor
- 27. Classification of Axonal Flow • Slow Transport – Antegrade, 0.1 to 4 mm/day • Fast Transport – Antegrade at up to 400 mm/day
- 31. Classification of Axonal Flow • Slow Transport – Antegrade, 0.1 to 4 mm/day • Fast Transport – Antegrade at up to 400 mm/day – Retrograde at 40-400 mm/day
- 33. Slow Axonal Transport: ~1-4 mm/day Delivery of cytosolic and cytoskeletal proteins to the nerve terminal: Microtubules, Neurofilaments, Enzymes
- 34. The Cytoskeletons of Neurons and Glia (and all eukaryotic cells!) Microtubules (Tubulin)Tubulins, MAPs, Motors: Kinesins and Dyneins Microfilaments (Actin)Actins, Actin Monomer Binding Proteins, Capping Proteins, Gelsolin Family, Crosslinking and Bundling Proteins, Tropomyosin, Motors: Myosin Intermediate Filaments- Superfamily of 5 classes: Types I and II: Keratins, Type III: GFAP, Vimentin, Desmin, Peripherin, Type IV: NF Triplet, Internexin, Nestin, Type V: Nucelar Laminins
- 36. The Substrate • Microfilaments • Microtubules • The Package • ATP !!!
- 37. Microtubules Subunit: tubulin MW: ~50 kD, α- é s β-tubulin -> heterodimer 1 bound GTP or GDP; α β
- 38. Microtubules
- 42. Requires ATP/Mg2+ as fuel for the motor
- 43. Road Map for the Session • • • • • • • Introduction and need for this discussion Historical aspects and the pioneers Characterization of the types of axoplasmic flow The molecular “motors” Integration of concepts Clinical utilization of the information – pathogenesis Clinical utilization of the information – therapeutics • Further reading
- 44. Kinesins
- 47. Kinesins Kinesins are a large family of proteins with diverse structures. Mammalian cells have at least 40 different kinesin genes. The best studied is referred to as conventional kinesin, kinesin I, or simply kinesin. Some are referred to as kinesin-related proteins (KRPs). Kinesin I has a structure analogous to but distinct from that of myosin. There are 2 copies each of a heavy chain and a light chain.
- 48. C-terminal tail domains light chains Kinesin I stalk domain hinge N-terminal heavy chain motor domains (heads)
- 50. Single kinesin moving a bead
- 51. Kinesin superfamily proteins (KIFs) bind to cargoes through adaptor or scaffolding protein complexes.
- 52. Kinesin superfamily proteins (KIFs) and cargoes for axonal and dendritic transport.
- 53. Rafts and cytoskeletal proteins as new cargoes Molecular motors: from one motor many tails to one motor many tales. Lawrence S.B. Goldstein Trends in Cell Biology, 2001, 11:12:477-482
- 55. Walking along the microtubules Single Headed Kinesin…
- 56. Kinesin Inactivation scaffolding protein microtubule kinesin cargo vesicle receptor inactive kinesin
- 57. So, how does it all work together?
- 58. Fast Axonal Transport: 100-400 mm/day Purpose: Transport organelles such as mitochondira and vesicles carrying SV and plasma membrane proteins to the nerve terminal. Also retrograde movement of vesicles containing neurotrophic factors back to the cell body.
- 59. Dyneins
- 60. The dynein microtubule motor. Biochim Biophys Acta. 2000. 1496:60-75.
- 61. Road Map for the Session • • • • • • • Introduction and need for this discussion Historical aspects and the pioneers Characterization of the types of axoplasmic flow The molecular “motors” Integration of concepts Clinical utilization of the information – pathogenesis Clinical utilization of the information – therapeutics • Further reading
- 63. Summary for axoplasmic transport • • • • Necessity Types Kinesins Dyenins • Summation • Need for this information !!!
- 64. Road Map for the Session • • • • • • • Introduction and need for this discussion Historical aspects and the pioneers Characterization of the types of axoplasmic flow The molecular “motors” Integration of concepts Clinical utilization of the information – pathogenesis Clinical utilization of the information – therapeutics • Further reading
- 66. • • Axonopathy and transport deficits early in the pathogenesis of Alzheimer's disease. Stokin GB, Lillo C, Falzone TL, Brusch RG, Rockenstein E, Mount SL, Raman R, Davies P, Masliah E, Williams DS, Goldstein LS Science 2005 Feb 25; 307(5713):1282-8
- 67. • Selective vulnerability and pruning of phasic motoneuron axons in motoneuron disease alleviated by CNTF. Pun S, Santos AF, Saxena S, Xu L, Caroni PNat Neurosci 2006 Mar 9(3):408-19 • Charcot-Marie-Tooth disease type 2A caused by mutation in a microtubule motor KIF1Bbeta. Zhao C, Takita J, Tanaka Y, Setou M, Nakagawa T, Takeda S, Yang HW, Terada S, Nakata T, Takei Y, Saito M, Tsuji S, Hayashi Y, Hirokawa NCell 2001 Jun 1 105(5):587-97
- 68. • 1-Methyl-4-phenylpyridinium induces synaptic dysfunction through a pathway involving caspase and PKCdelta enzymatic activities. Proc Natl Acad Sci U S A. 2007 Feb 13;104(7):2437-41 – Model for neurodegenration
- 69. • Jones LG, Prins J, Park S, Walton JP, Luebke AE, Lurie DI. • beading, and temporal processing deficits within the murine auditory brainstem. J Comp Neurol. 2008 Feb 20;506(6):1003-17. • Pan T, Kondo S, Le W, Jankovic J. • Lead exposure during development results in increased neurofilament phosphorylation, neuritic • The role of autophagy-lysosome pathway in neurodegeneration associated with disease. • Brain. 2008 Jan 10; [Epub ahead of print] Parkinson's
- 70. • Inflammation, demyelization,neurodegeneration, and neuroprotection in the pathogenesis of mutliple sclerosis. Peterson, Lisa K. , Fujinami, Robert S. • • • Journal Neuroimmunology 184 (2007): 37-44 multiple sclerosis: Role in symptom production, Sodium channels and damage and therapy. Smith, Kenneth J. Brain Pathology 2007 Apr;17(2):230-42.
- 71. • • Proteomic analysis of rat cortical neurons after fluoxetine (FLUX) treatment Long-Term Impairment of Anterograde Axonal Transport Along Fiber Projections Originating in the Rostral Raphe Nuclei After Treatment With Fenfluramine or Methylenedioxymethamphetamine (MDMA)
- 72. Is it only bad news ?
- 73. Road Map for the Session • • • • • • • Introduction and need for this discussion Historical aspects and the pioneers Characterization of the types of axoplasmic flow The molecular “motors” Integration of concepts Clinical utilization of the information – pathogenesis Clinical utilization of the information – therapeutics • Further reading
- 77. Road Map for the Session • • • • • • • Introduction and need for this discussion Historical aspects and the pioneers Characterization of the types of axoplasmic flow The molecular “motors” Integration of concepts Clinical utilization of the information – pathogenesis Clinical utilization of the information – therapeutics • Further reading
- 78. Further Reading
Hinweis der Redaktion
- Rabies does not wait, replicate in the cell body immediately and come back..
- Figure 4 | Kinesin superfamily proteins (KIFs) bind to cargoes through adaptor or scaffolding protein complexes. a | KIF13A binds to β1-adaptin of the AP1 (adaptor protein 1) adaptor complex and the AP1 adaptor complex binds to the mannose-6-phosphate receptor (M6PR)80. The AP1 adaptor complex comprises the β1-, γ-, 1- and δ1-adaptin subunits. β1-adaptin has three domains — trunk, hinge and ear — and the carboxy (C)-terminal tail of KIF13A binds to the ear domain. b | the C-terminal tail of KIF17 binds to one of the PDZ domains of LIN10 (Munc18-interacting protein, MINT1)65. LIN10, LIN2 (CASK, calcium/calmodulin dependent serine protein kinase) and LIN7 (VELIS, vertebrate LIN7 homologue/MALS, mammalian LIN7 protein), all have PDZ domains and interact through regions other than the PDZ domains to form a tripartite scaffolding protein complex, which binds to the NR2B subunit of NMDA (N-methyl-D-aspartate) receptors.
- Figure 3 | Kinesin superfamily proteins (KIFs) and cargoes for axonal and dendritic transport. a | A typical neuron, extending several dendrites (left) and a single thin axon (right) from the cell body. In the axon, microtubules are unipolar, with the plus ends pointing towards the synaptic terminal. Microtubules form special bundles at the initial segment, which might serve as the cue for axonal transport. Tubulovesicular organelles are transported anterogradely along microtubules by KIFs. In the growth cone of an axon collateral, KIF2A controls microtubule dynamics and the extension of collaterals. Rough endoplasmic reticula are abundant in most parts of the cell body, except for the axon hillock. Dendrites contain some rough endoplasmic reticlula. Microtubules have mixed polarity in proximal dendrites, but are unipolar in distal dendrites, with the plus end pointing away from the cell body. Membranous organelles and RNA-containing granules are transported along microtubules by KIFs. b | KIF5 transports vesicles containing APP (amyloid precursor protien) and APOER2 (apolipoprotein E receptor 2) by interacting with KLC (kinesin light chain)46,47,51,96,97. Mitochondria are transported by KIF5 and KIF1Bα27,45. KIF3 transports vesicles associated with fodrin57. KIF1A and KIF1Bβboth transport synaptic vesicle precursors26,31,32. JIPs, scaffolding proteins of the c-Jun amino (N)-terminal kinase (JNK) signalling pathway; KAP3, kinesin superfamily-associated protein 3. c | In dendrites, KIF5 transports vesicles containing AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid) receptors through an interaction between KIF5 and GRIP1 (glutamate receptor-interacting protein 1)68. RNA-containing granules are also transported by interacting directly with KIF5 (REF. 77). KIF17 transports vesicles containing NMDA (N-methyl-D-aspartate) receptors by interacting through the LIN complex, a tripartite protein complex containing mammalian homologues of the Caenhorhabditis elegans presynaptic density zone (PDZ) proteins LIN-2, LIN-7 and LIN-1065.
- Different general proposed mechanisms for attachment of kinesin motors to 'cargos'. Comparable mechanisms have also been proposed for dynein motors, but, for simplicity, only kinesins are shown. Motor proteins might link directly to transmembrane proteins, to scaffold proteins that link to transmembrane proteins, to proteinaceous raft complexes that bind to other protein cargos or possibly directly to other cytoskeletal filaments. How the dynactin complex, which in some cases might link dynein to spectrin networks on vesicles, fits into these mechanisms is unclear. See main text for additional details.
- Fig. 1. Dynein architecture and heavy chain organization. (a) Model of a generic two-headed dynein particle indicating the general location of various structural and functional domains described in the text. (b) Map of the dynein heavy chain identifying the segments of these large proteins involved in binding the IC/LC complex, ATP and microtubules.
