The determination of point groups
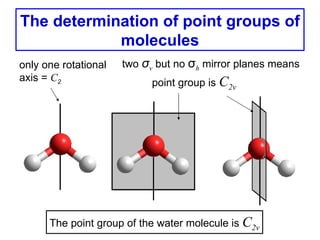
- 1. The determination of point groups of molecules only one rotational two σv but no σh mirror planes means axis = C2 point group is C2v The point group of the water molecule is C2v
- 2. Naming point groups: The name of the point group has information about the symmetry elements present. The letter is the rotational group and the subscript number after the letter indicates the order of the principal rotational axis (e.g. 3-fold or 4 fold etc.): A ‘D’ indicates an n-fold principal rotation axis A ‘C’ indicates only plus n 2-fold axes at one rotational axis right angles to it C3 C3v D4d D4h 3-fold rotational has σv but 4-fold d = no ‘h’ indicates axis no σh mirror principal σh mirror a σh mirror planes in a C group axis plane plane
- 3. Naming point groups (contd.): A subscript ‘h’ means that there is a σh mirror plane at right angles to the n-fold principal axis: C4 principal axis C3 principal axis only one of the three σv planes is shown σh σv D4h D3d A subscript ‘d’ (or v for C groups) means there is no σh mirror plane, but only n σ mirror planes containing the principal C axis.
- 4. Naming platonic solids: Platonic solids: T = tetrahedral = 4 three-fold axes O = octahedral = 3 four-fold axes I = icosahedral = 6 five-fold axes C60 ‘bucky-ball’ or ‘Fullerene’ Td Oh Ih
- 5. Flow chart for determining point groups.
- 6. The point group of the carbon dioxide molecule i We start at the top of the flow-chart, and can see that C∞ the CO2 molecule is linear, and has a center of inversion (i) so it is D∞h. Note the C∞ D∞h principal rotation axis.
- 7. Other linear molecules: The top row of linear molecules all have a center of inversion (i) and so are D∞h. i D∞h i N2 O2 F2 H2 HC≡N HI C≡O The bottom row have no C∞v All have a C∞ i and so are C∞v axis
- 8. The Platonic solids: tetrahedron octahedron icosahedron Td Oh Ih C60 ‘buckyball’
- 9. The Cs point group: I σ Cs C Cl chloro-difluoro-iodo- F methane F
- 10. Most land animals have bilateral symmetry, and belong to the Cs point group: Cs Mirror planes (σ) Cs
- 11. The C1 point group: Molecules that have no symmetry elements at all except the trivial one where they are rotated through 360º and remain unchanged, belong to the C1 point group. In other words, they have an axis of 360º/360º = 1-fold, so have a C1 axis. Examples are: I I N Cl C H Br Cl C1 F C1 Bromo-chloro-fluoro-iodo- chloro-iodo-amine methane
- 12. The division into Cn and Dn point groups: After we have decided that there is a principal rotat- ional axis, we come to the red box. If there are n C2 axes at right angles to the principal Dn axis, we have a Dn point group, If not, it is a Cn Cn point group.
- 13. The Cn point groups: The Cn point groups all have only a single rotational axis, which can theoretically be very high e.g. C5 in the complex [IF6O]- below. They are further divided into Cn, Cnv, and Cnh point groups. C5 •The Cn point groups have no other symmetry elements •the Cnv point groups have also n mirror planes containing the Cn rotational axis •the Cnh point groups also have a σh mirror plane at right angles to the principal rotational axis. [IF6O]-
- 14. The point group of the water molecule We start at the top of the flow-chart, and can see that the water molecule is not linear, and is not tetrahedral (Td), octahedral (Oh), or icosahedral, (Ih) so we proceed down the chart
- 15. C2 Yes, there is a principal Cn axis, so we proceed down the chart, but in answer to the next question, there are no further C2 axes at right angles to the principal axis, which is the only axis, so we proceed down the chart
- 16. C2 C2 C2 there is no σh plane at right angles to σv σv the C2 axis, but there are two σv planes containing the C2 axis. The point group of the water molecule is C2v
- 17. Other Cnv molecules: water ammonia σv C2v C3v σv C4v V σv Vanadyl tetrafluoride (VOF4)
- 18. Some more C2v molecules: C2 σv C2 σv C2 σv P S C σv σv σv Phosphorus iodo- sulfur tetra- carbonyl tetrafluoride (PF4I) fluoride (SF4) chloride (COCl2)
- 19. The Cn point groups: These have a Cn axis as their only symmetry element. They generally resemble propellers which have the front and back different. Important examples are (hydrogens omitted for clarity): triphenyl C3 Cobalt(III) phosphine tris-glycinate viewed down viewed down C3 axis C3 C3 axis C3 C3 C3 C3 triphenyl Cobalt(III) phosphine tris-glycinate viewed from viewed from the side the side
- 21. The Dnh point groups: C4 four C2 mirror plane principal axes at at rt. angles rt. angles axis to C4 axis to C4 axis C2 C2 σh C2 C2 D4h
- 22. Examples of molecules belonging to Dnh point groups: C2 C3 C3 C3 D2h D3h D3h D3h C4 C5 C4 C5 D4h D4h D5h D5h
- 23. Benzene, an example of the D6h point group: C6 C2 principal axis C2 C2 C6 σh C2 σv σv D6h C6 principal axis C6 principal axis
- 24. The Dn point groups: these have a principal n-fold axis, and n 2-fold axes at right angles to it, but no mirror planes. C2 principal axis C N C2 [Cu(en)2]2+ complex Cu N with H-atoms omitted for clarity. (en = ethylene diamine) C2 D2
- 25. Some further views of the symmetry elements of [Cu(en)2]2+, point group D2 : C2 principal axis [Cu(en)2]2+ complex C2 D2 with H-atoms omitted for clarity. (en = ethylene diamine) C2 principal C2 axis C2 principal C2 principal C2 axis axis C2 C2 C2 C2 C2
- 26. Some views of the symmetry elements of [Co(en)3]3+, point group D3. C2 C3 principal axis C2 C2 C3 D3 C2 principal axis axis view down the C3 axis view down one of the of [Co(en)3]3+ showing three C2 axes of [Co(en)3]3+ the three C2 axes. at right angles to C3
- 27. Other examples of the D3 point group C3 C2 C2 C2 principal axis C2 C2 C2 D3 D3 [Co(oxalate)3]3- [Co(bipyridyl)3]3+
- 28. Some cobalt(III) complexes belonging to the D3 point group: C2 C2 C2 CH3 3+ 3+ NH2 H H3C O CH3 N 2 H 2N N O O N N Co Co Co O O H 2N N N N H2 H3C O CH3 NH2 N CH3 tris(ethylenediamine) tris(2,2’-bipyridyl) tris(acetylacetonato) cobalt(III) cation cobalt(III) cation cobalt(III) D3
- 29. Comparison of C3 and D3 tris(chelates) no C2 axis at this point C2 D3 O C3 3+ O H2 NH2 H 2 H 2N N H2N N Co Co O O H2N N O H2 NH2 NH2 O tris(ethylenediamine)cobalt(III) tris(glycinato)cobalt(III)
- 30. Molecules belonging to the Dnd point groups These have mirror planes parallel to the principal axis, but not at right angles to it. C5 axis C3 axis σv planes contain the principal axis D3d D5d Staggered form of ethane Staggered form of ferrocene
- 31. The D4d point group: C4 principal axis σv C2 C2 σv σv C2 C2 C2 σv C4 C4 [ZrF8] 4- principal axis Square antiprism principal axis D4d As predicted by VSEPR, the [ZrF8]4- anion has a square anti-prismatic structure. At left is seen the C4 principal axis. It has four C2 axes at right angles to it, so it has D4 symmetry. One C2 axis is shown side-on (center). There are four σv mirror planes (right), but no mirror plane at
- 32. [K(18-crown-6)]+, an example of a D3d point group: C3 principal axis C3 principal axis σv C2 C2 K+ σv C2 C2 C2 C2 σv D3d The complex cation [K(18-crown-6)]+ above is an important structure that has D3d symmetry. It has a C3 principal axis with 3 C2 axes at right angles to it, as well as three σv mirror planes that contain the C3 axis, but no σh mirror plane (because it’s not flat, as seen at center), so is D3d.